
Clarivate oncology experts analyzed ASCO 2021 abstracts from Mainland China to highlight key trends and breakthrough treatments that are expected to impact the oncology landscape.
Mainland Chinese companies were strongly represented at the 2021 ASCO Annual Meeting. As part of our Drugs to Watch™ series spotlighting therapies expected to have significant impact on drug landscapes, Clarivate China In-Depth™ experts reviewed more than 200 abstracts from Mainland China and identified a few notable trends, including:
- A sharp increase in the number of oral presentations from companies in Mainland China from 11 at ASCO 2020 to 20 at ASCO 2021
- Increased representation of Chinese companies posting, encouraging results from pivotal clinical trials of innovative therapies across solid tumor types.
Figure 1: Number of oral presentations from Mainland China at ASCO
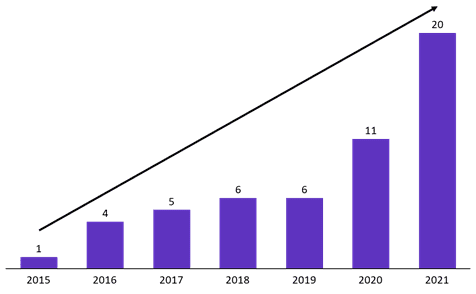
Source: ASCO meeting library
1. Abemaciclib took the spotlight, showing benefit in lowering the recurrence rate in high-risk early-stage HR-positive/HER2-negative breast cancer
Abemaciclib (Eli Lilly’s Verzenio) is set to become the first CDK4/6 inhibitor in Mainland China to gain approval for HR-positive/HER2-negative, high-risk early breast cancer1
Context: No drug therapy is currently indicated for reducing the risk of recurrence of the early-stage HR-positive/HER2-negative breast cancer in Mainland China. Multiple CDK4/6 inhibitors are being evaluated as adjuvant therapies for this high-risk breast cancer patient subpopulation.
What’s new at ASCO: Eli Lilly presented results from the Chinese patient subpopulation enrolled in the pivotal Phase III MonarchE trial—evaluating abemaciclib with standard adjuvant endocrine therapy (ET) versus ET alone in patients with HR-positive/HER2-negative early-stage breast cancer. The CDK4/6 inhibitor not only showed benefit in the primary endpoint of invasive disease-free survival, but also in distant relapse-free survival. Its safety profile in Chinese patients was consistent with that observed in global populations.
What to expect: The results are highly encouraging and will likely support the label expansion of abemaciclib as adjuvant therapy for high-risk, early-stage HR-positive/HER2-negative breast cancer following surgery. In Mainland China, this would enable Eli Lilly to have a significant first-mover advantage within a highly competitive space, where many other CDK4/6 inhibitors (e.g., Jiangsu Hengrui Medicine’s dalpiciclib) are being developed but are far behind abemaciclib.
2. Dalpiciclib may challenge other CDK4/6 inhibitors in the recurrent metastatic HR-positive/HER2-negative breast cancer market for patient share
Chinese physicians will welcome a novel treatment option for this difficult-to-treat patient population2
Context: In December 2020, Jiangsu Hengrui Medicine announced that dalpiciclib (SHR6390) met its primary endpoint in a pivotal Phase III (DAWNA-1) clinical study in recurrent metastatic HR-positive/HER2-negative breast cancer. Based on these results, Jiangsu Hengrui Medicine filed an NDA for dalpiciclib combined with fulvestrant for this treatment setting in Mainland China and gained priority review status in April 2021.
What’s new at ASCO: Jiangsu Hengrui Medicine presented results from the pivotal Phase III (DAWNA-1) study. The data presented at ASCO demonstrate that dalpiciclib in combination with fulvestrant significantly improves progression free survival (PFS) for this patient segment over placebo plus fulvestrant, and demonstrates a manageable safety profile.
What to expect: If approved, dalpiciclib will become the third CDK4/6 inhibitor—after Pfizer’s palbociclib in July 2018 and Eli Lilly’s abemaciclib in January 2021—to be approved by the NMPA for this patient population. Considering the meaningful improvement that the CDK4/6 inhibitors (combined with fulvestrant) have shown in delaying disease progression in recurrent metastatic breast cancer patients, the drug class could soon emerge as a preferred treatment option in this setting.
3. Can pyrotinib/trastuzumab challenge the status of pertuzumab/trastuzumab as the preferred neoadjuvant therapy for HER2-positive breast cancer patients?
Although pyrotinib (Aruini) shows early positive signs of efficacy, more data are required to analyze its full potential in the neoadjuvant treatment setting3
Context: Jiangsu Hengrui Medicine is investigating pyrotinib (combined with trastuzumab and chemotherapy) as an adjuvant therapy in early-stage HER2-positive breast cancer in Mainland China.
What’s new at ASCO: Jiangsu Hengrui Medicine presented results from a Phase II study of pyrotinib as adjuvant therapy in early-stage HER2-positive breast cancer. Results showed that pyrotinib combined with trastuzumab and chemotherapy significantly improves the total pathologic complete response (tpCR) rate, by approximately two times compared with the trastuzumab plus chemotherapy regimen. It also shows a manageable safety profile in this patient population.
What to expect: In addition to tpCR (the primary outcome measure), data on secondary outcome measures (e.g., event-free survival, disease-free survival, distant disease-free survival and objective response rate) will be critical in appropriately evaluating its clinical efficacy. The company is also conducting a pivotal Phase III clinical trial in Mainland China to assess the safety and efficacy of pyrotinib plus trastuzumab with chemotherapy versus trastuzumab with chemotherapy. We anticipate that if the trial shows positive results for pyrotinib plus trastuzumab, the regimen will challenge the pertuzumab plus trastuzumab regimen for market share in the neoadjuvant treatment setting.
4. Aumolertinib proves effective in treating EGFR-Positive, first-line metastatic NSCLC
Positive Phase III data will allow aumolertinib (Ameile) to emerge as a contender to the only other third-generation EGFR inhibitor, osimertinib (Tagrisso), in the first-line metastatic non-small cell lung cancer (NSCLC) segment4
Context: Based on positive Phase III results announced in February 2021, Hansoh Pharma received breakthrough therapy designation in April 2021 and priority review status in May 2021 for its innovative third-generation EGFR inhibitor aumolertinib for the treatment of first-line metastatic patients with EGFR exon 19 del or L858R mutations in Mainland China.
What’s new at ASCO: The pivotal Phase III clinical trial results showed that aumolertinib significantly prolongs PFS and duration of response (DoR) compared with gefitinib and has a favorable safety profile.
What to expect: If approved, aumolertinib will be only the second innovative third-generation EGFR inhibitor in the first-line metastatic NSCLC segment in Mainland China, following Tagrisso’s approval in September 2019. Aumolertinib’s approval will further boost the use of third-generation EGFR inhibitors in the country; however, Tagrisso’s first-to-market advantage, high affordability and the availability of its generics will limit the uptake of aumolertinib in this treatment setting.
5. Donafenib shows superiority over Sorefenib (Nexavar) in first-line advanced hepatocellular carcinoma
Is positive Phase III data enough for donafenib to experience substantial uptake in the first-line advanced hepatocellular carcinoma (HCC) setting?5
Context: Sorafenib has long been the unrivaled standard of care as a first-line treatment of advanced-stage HCC across the globe. However, it offers only modest overall survival (OS) benefits and is associated with serious adverse events. A Phase III study to evaluate lenvatinib (REFLECT) in this treatment setting also failed to demonstrate a statistically significant improvement in OS versus sorafenib. Therefore, an unmet need remains for agents that offer improved overall survival benefit and safety over existing treatment options in this patient population.
What’s new at ASCO: Zelgen Biopharmaceuticals presented the results from a head-to-head pivotal Phase III study of donafenib versus sorafenib in first-line advanced HCC patients. Results showed that donafenib significantly improves the OS of patients over sorafenib and has a favorable safety and tolerability profile.
What to expect: Although encouraging, donafenib’s survival benefit over sorafenib is expected to be overshadowed by the likely emergence of combination therapies pairing immune checkpoint inhibitors with TKIs, VEGF inhibitors, and CTLA-4 inhibitors. Such combination therapies are being developed by several drug manufacturers for the Chinese HCC market. Many of these therapies are being investigated in late-phase clinical studies for first-line advanced HCC and are expected to offer superior efficacy and tolerability over TKIs alone in this treatment setting. Donefenib’s prospects of a meaningful market uptake in the first-line treatment setting will also be challenged by sorafenib’s long market presence leading to high physician comfort, high affordability and the availability of cost-effective generics.
6. Toripalimab shows promise as first-line treatment for recurrent or metastatic nasopharyngeal carcinoma
Positive results will support the label expansion of Toripalimab (Junshi Biosciences’ Tuoyi) in the first-line metastatic nasopharyngeal carcinoma (NPC) patient population, and will further cement the growing dominance of domestic manufacturers in Mainland China’s PD-1 inhibitor market 6
Context: In February 2021, Junshi Biosciences filed a supplemental NDA for toripalimab (combined with chemotherapy) as a first-line treatment for advanced metastatic NPC in previously untreated patients. The filing was based on positive Phase III JUPITER-02 clinical trial results announced in September 2020.
What’s new at ASCO: Junshi Biosciences presented results from the Phase III JUPITER-02 clinical trial, showing that toripalimab with gemcitabine/cisplatin as first-line treatment provides superior PFS and objective response rate (ORR), along with a longer DOR, versus gemcitabine/cisplatin alone and shows a manageable safety profile.
What to expect: Toripalimab is already approved for second-line treatment of NPC, and the label expansion will allow it to become the second PD-1 inhibitor, after camrelizumab (AiRuiKa) (approved in June 2021), for the first-line treatment of recurrent or metastatic NPC – a setting with high unmet need and largely untapped market potential within Mainland China. However, despite being included in the National Reimbursement Drug List, toripalimab will face fierce competition from camrelizumab and the upcoming PD-1 inhibitors in Phase III studies for NPC, including tislelizumab (Baizean) and sintilimab (Daboshu).
These results and continued progress in the field are a testament to the growing influence of Mainland China’s domestic manufacturers, beyond the country’s PD-1 inhibitor market and now globally. With FDA’s breakthrough designation for treatment of NPC, Tuoyi represents an international leap for therapies developed in Mainland China.
Clarivate oncology experts who contributed to this article include Amardeep Singh, Akash Saini, Gang Wang and Shuxin Bao.
To learn more about our take on other oncology treatment landscape game changers, follow our ASCO 2021 analysis here.
References
- Efficacy and safety analysis of Chinese patients in monarchE: Abemaciclib combined with adjuvant endocrine therapy for high-risk HR+, HER2- early breast cancer. ASCO 2021. Abstract 522.
- Dalpiciclib versus placebo plus fulvestrant in HR+/HER2- advanced breast cancer that relapsed or progressed on previous endocrine therapy (DAWNA-1): A multicenter, randomized, phase 3 study. ASCO 2021. Abstract 1002.
- Pyrotinib as neoadjuvant therapy for HER2+ breast cancer: A multicenter, randomized, controlled, phase II trial. ASCO 2021. Abstract 574
- Randomized phase III trial of aumolertinib (HS-10296, Au) versus gefitinib (G) as first-line treatment of patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) and EGFR exon 19 del or L858R mutations (EGFRm). ASCO 2021. Abstract 9013.
- Donafenib versus sorafenib as first-line therapy in advanced hepatocellular carcinoma: An open-label, randomized, multicenter phase II/III trial. ASCO 2021. Abstract 4506.
- JUPITER-02: Randomized, double-blind, phase III study of toripalimab or placebo plus gemcitabine and cisplatin as first-line treatment for recurrent or metastatic nasopharyngeal carcinoma (NPC). ASCO 2021. Abstract LBA2.









