
Major improvements in the regulatory environment as well as changes in strategies of multinational companies have led to a general decrease in the time to marketing authorization, improved consistency, as well as an increase in the number of medicines that have become available over the last decade, 2009-2018, across six major regulatory agencies.
These are some of the findings analyzing regulatory approval of new active substances (NASs) from a new study released by the Centre for Innovation in Regulatory Science (CIRS) in R&D Briefing 70, titled “New drug approvals in six major authorities 2009-2018: Focus on Facilitated Regulatory Pathways and Orphan Status.”
The six agencies included the European Medicines Agency (EMA), the U.S. Food and Drug Administration (FDA), the Japan Pharmaceuticals and Medical Devices Agency (PMDA), Health Canada, Swissmedic and the Australian Therapeutic Goods Administration (TGA) (See Figure 1).
CIRS analysis shows that the number of common products approved by all six agencies increased from 16 in 2009-2013 to 52 in 2014-2018, which indicates that more products were becoming internationalized.
Underlying factors influencing the overall time it takes for a new medicine to be submitted and then approved by an agency include company strategy, the conduct and the type of the review process, the type of the product and its therapeutic area. More specifically, facilitated regulatory pathways (FRPs), orphan drug designation and sponsor size are major elements of the submission and approval strategies.
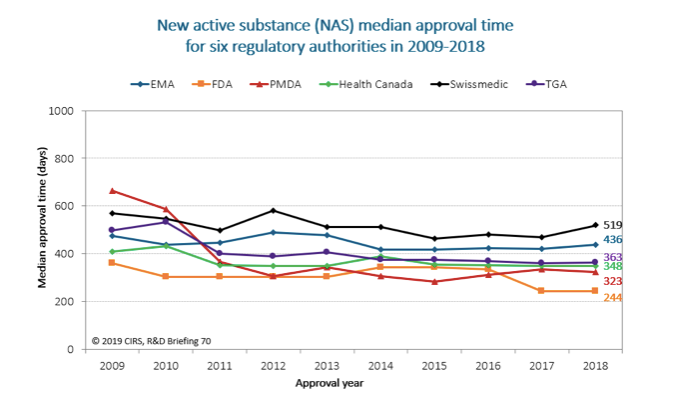
Figure 1. Improvements in the regulatory environment have helped lead to a general decrease in the time to marketing authorization.(Approval time is calculated from the date of submission to the date of approval by the agency. This time includes agency and company time. EMA approval time includes the EU Commission time.) Source: Centre for Innovation in Regulatory Science
Focus on facilitated regulatory pathways
All six agencies now offer one particular FRP, namely an expedited review (referred to by the EMA as an “Accelerated Assessment,” by Swissmedic as “Fast-Track” and by the other four agencies as “Priority Review”) designed to hasten the review process of promising new active substances. TGA implemented its priority system in 2017 and first approvals were granted in 2018. In general, expedited reviews were approved faster than standard ones.
In 2018, expedited approvals accounted for the highest proportion of approvals for FDA (73%), followed by Health Canada (35%), PMDA (28%), Swissmedic (13%), EMA and TGA (10%).
Out of the six agencies, FDA offered (or made available) the greatest number of FRPs to enable the availability, review and/or approval of medicines where there is an unmet medical need. In 2018, 75% of NASs approved by FDA benefitted from at least one of the available FRPs (orphan excluded). At the other agencies, FRPs ranged from 10% for TGA to 41% for Health Canada. (See Figure 2.)
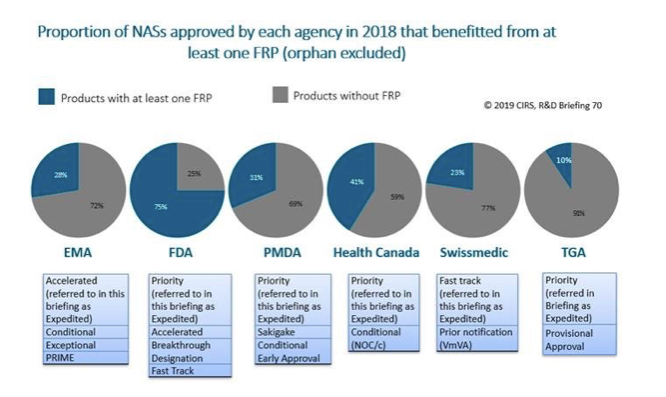
Figure 2. Out of the six agencies, the FDA offered (or made available) the greatest number of FRPs to enable the availability, review and/or approval of medicines where there is an unmet medical need. Source: Centre for Innovation in Regulatory Science.
Focus on orphan status
The number of NASs with an orphan designation has increased across the six agencies, from 25% in 2009-2013 to 38% in 2014-2018. In 2018, FDA had the highest approval number for orphans (35) while PMDA had the lowest (eight). Health Canada does not currently have an orphan policy; this agency however approved 15 NASs in 2018 that were classified as orphan by either FDA, EMA or TGA.
FDA had the fastest median approval time for orphans in 2018 (243 days), 88% of them being approved through expedited review. Among the six agencies, EMA was the only one with median approval time longer for orphans than for non-orphans. For Swissmedic these times were similar in 2018. At TGA, 20% of orphan drugs were approved in 2018 with the newly introduced priority review.
Out of the 52 NASs approved by all six authorities in 2014-2018, only 10 NASs received an orphan designation across all the authorities, highlighting that there may be differences in criteria for obtaining the designation in each agency. In general, the median submission gap for an orphan NAS was longer compared to a non-orphan NAS.
The majority of orphan NASs were approved by non-top companies, highlighting the important role of smaller companies to drive innovation.
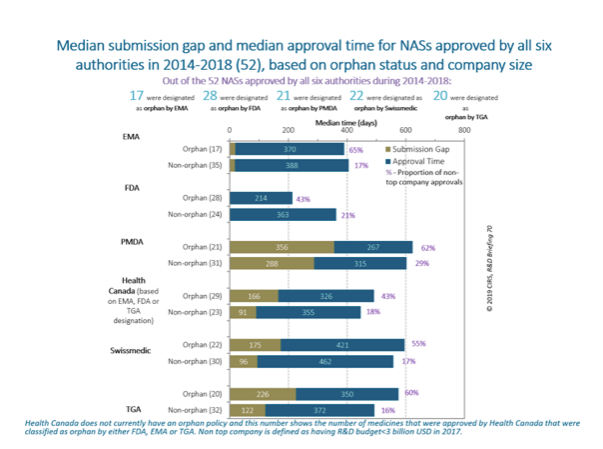
Figure 3. CIRS analysis showed that the majority of orphan NASs were approved by non-top companies, highlighting the important role of smaller companies to drive innovation. Source: Centre for Innovation in Regulatory Science.
Editor’s note: The full report from the author and her colleagues is available here from CIRS, the Centre for Innovation in Regulatory Science, the neutral, independently managed subsidiary company which forms part of Clarivate Analytics:



