DRG Fingertip Foundations
Pharmacist validated market access intelligence delivering coverage, restrictions, transparent policy sourcing, and policy change tracking.
Pharmacist-validated, AI-powered market access intelligence
Arming market access, forecasting, and commercial teams with actionable coverage, restriction, and policy intelligence across medical and pharmacy benefits—delivering a reliable foundation for access planning and execution.
Drive better payer engagement with actionable coverage clarity
Pharmacist‑validated coverage and policy intelligence sourced directly from health plans — giving your teams the confidence to plan faster, respond sooner, and execute with precision.

Reveal real-world accessbarriers
Identify real world access barriers with pharmacist validated coverage and restriction intelligence—so you can prioritize accounts, validate assumptions, and support payer strategy with confidence.

Turn policy into forecasting insight
Translate complex payer medical policies into standardized, analyst ready business rules that strengthen access assumptions and launch forecasts.

See your competitive position clearly
Compare policy criteria, restrictions, and benefit design across payers to reveal competitive strengths, uncover hidden hurdles, and fine‑tune your negotiation strategy.

Optimize access and payer strategy
Get coverage and restriction level clarity on medical and pharmacy benefits including prior authorization logic, step edits, sub indication criteria, and utilization management rules —empowering teams to anticipate restrictions, engage payers confidently, and prioritize access opportunities with clarity.
Foundational market access intelligence you can trust - validated by experts
The industry’s most accurate pharmacist validated coverage, restriction, and policy intelligence—built to support confident access decisions.
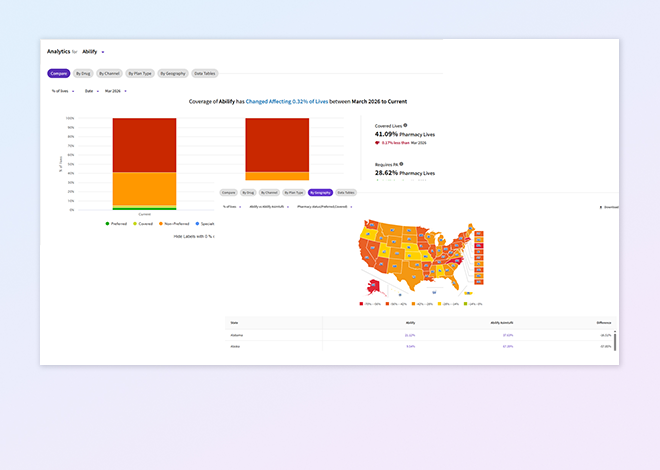
Fingertip coverage
Track real-world formulary coverage across medical and pharmacy benefit types, organized by plan, therapeutic area, and product to gain rapid visibility into your brand.
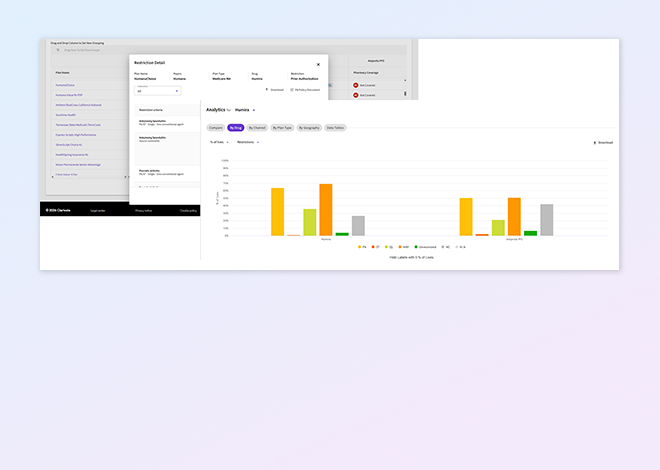
Fingertip restrictions
Reveal the restriction details that define access—prior authorization logic, step edits, sub indication criteria, and full coverage requirements—so you can see the true path to patient access.
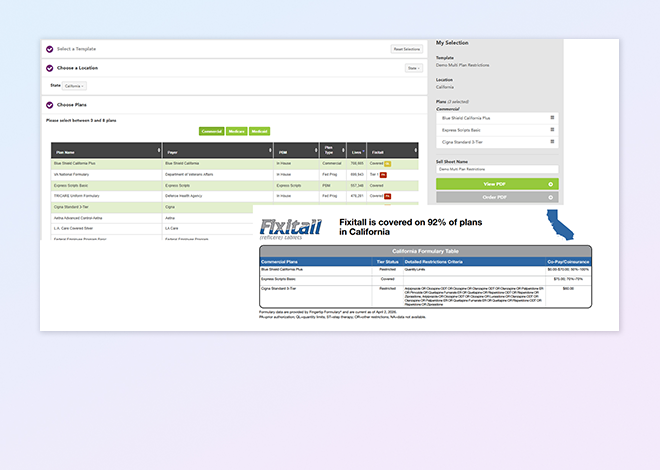
Fingertip publisher
Access complete policy documents—linked PDFs and structured metadata—showing exactly how payers define coverage, ensuring full transparency and compliance.
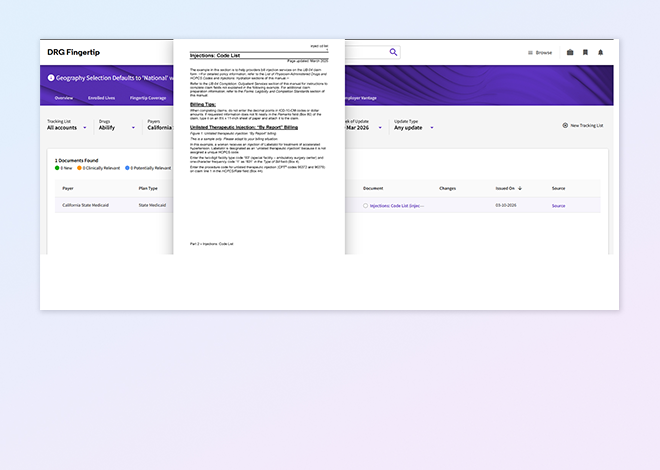
Fingertip PolicyTrack
Monitor payer policy changes over time with clear versioning and change alerts to support brand tracking and competitive monitoring.
Get trusted access clarity
Get definitive, pharmacist‑validated coverage and policy insights for your brand.
Questions we help you answer with confidence
Foundational market access questions teams must answer before strategy and execution.
- How is my brand covered compared to my competitors and what restrictions exist preventing access?
- How can I validate payer coverage assumptions to deliver credible forecasts that drive confident launch planning?
- How can I interpret payer medical policies to understand their impact on uptake and build actionable rules for forecasting and launch?
Why DRG Fingertip Foundations?
20+
15+ years
50+
Resources
Frequently asked questions
Market access coverage and restriction data describes how therapies are covered across medical and pharmacy benefits, including formulary status, prior authorization requirements, step edits, and policy criteria. This data helps teams understand real world access conditions and validate assumptions used in forecasting and payer strategy.
Fingertip combines AI‑powered extraction with clinical pharmacist validation and links every insight back to the original policy source for full transparency. The platform continuously refreshes data across 4,800+ formularies and 900+ payers, supported by more than 10 years of longitudinal data.
Policy changes are scraped and versioned across 950+ payer and plan types, with weekly email alerts highlighting new or updated policies. Teams can filter alerts by payer, book of business, document type, or update type to focus only on what matters.
Yes. Fingertip provides payer‑validated, sub‑indication‑level criteria, which can be translated into defensible business rules for forecast models. This helps teams reduce manual validation time, strengthen assumptions, and improve cross‑functional confidence in projections.
Yes. Fingertip insights can be tailored by indication, geography, benefit type, or internal taxonomy, and delivered via APIs, bridge files, or modular tools to fit your reporting, analytics, or field‑execution workflow.
Explore our market access solutions
Bring treatments to patients faster.










