
Accelerated innovation in drug design and development is providing diverse treatment options for cancer patients, including hematological malignancies, like multiple myeloma. However, innovation has complicated physicians’ therapy selection and decision making, including how best to sequence treatment across lines of therapy. Thereby contributing to an unmet need for indications like multiple myeloma, that are typically associated with frequent disease relapse and recurrence. Nevertheless, the pipeline for multiple myeloma is competitive and bursting with a potential arsenal of future treatment options. There are a few novel therapies that stand out as potential game changers and disruptors through 2020 and beyond.
Figure 1: Disruptors of the Multiple Myeloma Landscape

BCMA-targeted therapies on the rise
B-cell maturation antigen (BCMA), a cell-surface protein that is expressed highly on malignant plasma cells but has low expression on normal cells, is under intense research and development as a target for multiple myeloma therapies. Drug developers are pursuing novel and innovative approaches that target BCMA and disrupt its function. Broadly, these innovative approaches involve cell therapies or antibody-based therapeutics.
Figure 2: The clinical development landscape of key BCMA-target agents
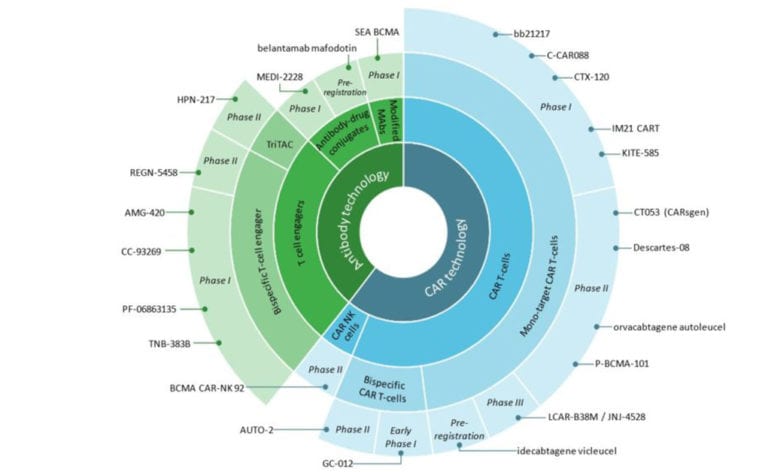
Novel cell therapy and antibody-based approaches emerge
Modified monoclonal antibodies, antibody-drug conjugates and bispecific T-cell engagers
- Monoclonal antibodies (MAbs): One strategy involves engineering non-fucosylated monoclonal MAbs that may elicit stronger antibody-dependent cytotoxicity that conventional MAbs (e.g. Seattle Genetics’ SEA-BCMA).
- Antibody drug conjugates (ADCs): ADCs target tumor cells and deliver a cytotoxic payload, preserving healthy cells; they are already revolutionizing treatments for non-Hodgkin’s lymphoma and breast cancer. The most advanced anti-BCMA ADC in development for multiple myeloma is GlaxoSmithKline’s belantamab mafodotin (GSK2857916). The agent secured FDA priority review in January 2020 following positive data from the pivotal DREAMM-2 study demonstrating a clinically meaningful improvement in overall response rate (ORR) (31%) in patients with heavily pretreated myeloma. It is expected to compete fiercely with BCMA-targeted CAR T-cell therapies.
- Bispecific T-cell engagers: These antibodies comprise one single-chain variable fragments (ScFv) that is directed against BCMA, and the other ScFv is directed against CD3 on cytotoxic T lymphocytes (CTLs). Key examples include Amgen’s BiTE AMG-420 and Bristol-Myers Squibb’s CC-93269.
- Trispecific antibodies: These antibodies may enhance therapeutic efficacy through T-cell receptor costimulation. Harpoon Therapeutics/AbbVie is developing HPN-217, a tri-specific T cell-activating construct (TriTAC). It is a recombinant fusion protein that combines an anti-CD3 ScFv with two single domain antibodies targeting BCMA and albumin.
Autologous CAR T-cell therapies, bispecific CAR T cells and CAR NK cells
Following the clinical success of anti-CD19 CAR T-cell therapies in other hematological malignancies, many companies are targeting BCMA using cell-based approaches.
- CAR T-cell therapies: Bristol-Myers Squibb/bluebird bio’s idecabtagene vicleucel (ide-cel; bb2121) is the clear CAR T-cell frontrunner in terms of clinical development, closely followed by Janssen/Legend Biotech’s JNJ-4528/LCAR-B38M. Based on the Phase II KarMMa study, a BLA for ide-cel was submitted to the FDA in March 2020. Ide-cel has achieved an impressive ORR of 73.4% (KarMMa). It is anticipated to face fierce competition from the BCMA-targeting ADC belantamab mafodotin in fourth- and later-lines owing to the ADC’s off-the-shelf manufacturing and ease of administration, which is expected to drive stronger uptake than ide-cel across the major markets.
- Bispecific CAR T-cell therapies: Some companies are working to further enhance target specificity with next-generation, dual-targeting bispecific CAR T-cell therapies. For example, Autolus’s AUTO2 that targets BCMA and TACI (transmembrane activator and calcium-modulator and cyclophilin ligand interactor), and Gracell Biotechnology’s CAR T cells that target BCMA and CD19.
- CAR NK cells: The feasibility of off-the-shelf cell therapies utilizing novel strategies such as CAR NK cells are being explored (e.g. Asclepius Technology’s BCMA CAR-NK 92 and/or Celularity’s PNK-007). Asclepius Technology’s allogeneic CAR NK cells are derived from human cell line NK-92, and unlike autologous CAR T-cell therapies, can be readily available because manufacturing is not dependant on an individual patient’s cells.
Darzalex continues to revolutionize treatment across lines
The anti-CD38 Mab Darzalex (Janssen/Genmab) was first approved as a monotherapy for the treatment of relapse or refractory (R/R) multiple myeloma patients (in 2015) and has since secured successive label expansions in this dynamic space. Darzalex-based regiments- (e.g., Darzalex combined with dexamethasone and either Revlimid, Velcade or Pomalyst/Imnovid) are now firmly entrenched in the R/R setting. Furthermore, positive data from the Phase III CANDOR trial is expected to solidify Darzalex’s label expansion as part of the triplet regimen DKd (Darzalex, Kyrpolis, and dexamethasone) in this patient population with Genmab/Janssen filing for FDA approval in February 2020.
Darzalex is also approved in the United States as part of key multi-drug regimens (e.g. D-VTD [Darzalex, Velcade, thalidomide, and dexamethasone] and D-VRD [Darzalex, Velcade, Revlimid, and dexamethasone]) for transplant-eligible and transplant-ineligible treatment-naïve patients, the first Mab to achieve approval in the first-line setting.
DRG forecast that Darzalex-based regimens will become key standards of care in the commercially lucrative first-line setting over the next decade. Darzalex’s uptake and revenue opportunity will also be driven by a future approval in patients with smoldering multiple myeloma (AQUILA study), launch of the more convenient subcutaneous formulation (PERSEUS and PLEIADES studies) and use of the drug as maintenance.
Future Market Considerations
Future prescribing and adoption of novel therapies for multiple myeloma will be dependent on many factors including clinical efficacy and safety, cost of therapy and patient access. Cost of therapy may impede patient access and in the United States, commercial or Medicare coverage will be pertinent to the therapies’ adoption. Over the next decade, DRG anticipates Darzalex-based regimens to dominate across treatment lines and become key standard of cares in earlier settings. Owing to these potential barriers as well as being largely restricted to late-line settings where there are fewer patients DRG forecasts BCMA-targeting approaches (CAR T-cell therapies and ADCs) to capture approximately 5% of total major market sales by 2028. However, given their anticipated premium pricing, these drug classes will garner substantially high sales figures. These novel therapies will address an important unmet need for patients who have received at least three prior lines of therapy and have limited therapeutic options remaining (particularly patients that are refractory to Darzalex).









