Discover the future of peripheral vascular devices
Medtech Insights Reports
Key trends in the PV device market:
- Uptake of premium-priced devices will drive growth.
- The launch of innovative devices for treating below-the-knee lesions will bolster the US PV device market going forward.
- Recently published positions and guidelines from key organizations are expected to impact the adoption of paclitaxel-eluting devices in Europe.
- Rising procedure volumes for iliac and lower limb indications and the increasing adoption of endovascular techniques over surgical intervention will also fuel market growth in the Asia Pacific region, particularly in China.
Request more information
The Peripheral vascular devices market insights reports provide comprehensive data and analysis on the state of the market for PV devices from 2019 through 2033. Backed by actionable market insights from Medtech experts, data, and unmatched research methodology the reports allow you to answer key questions and make data-driven portfolio decisions.
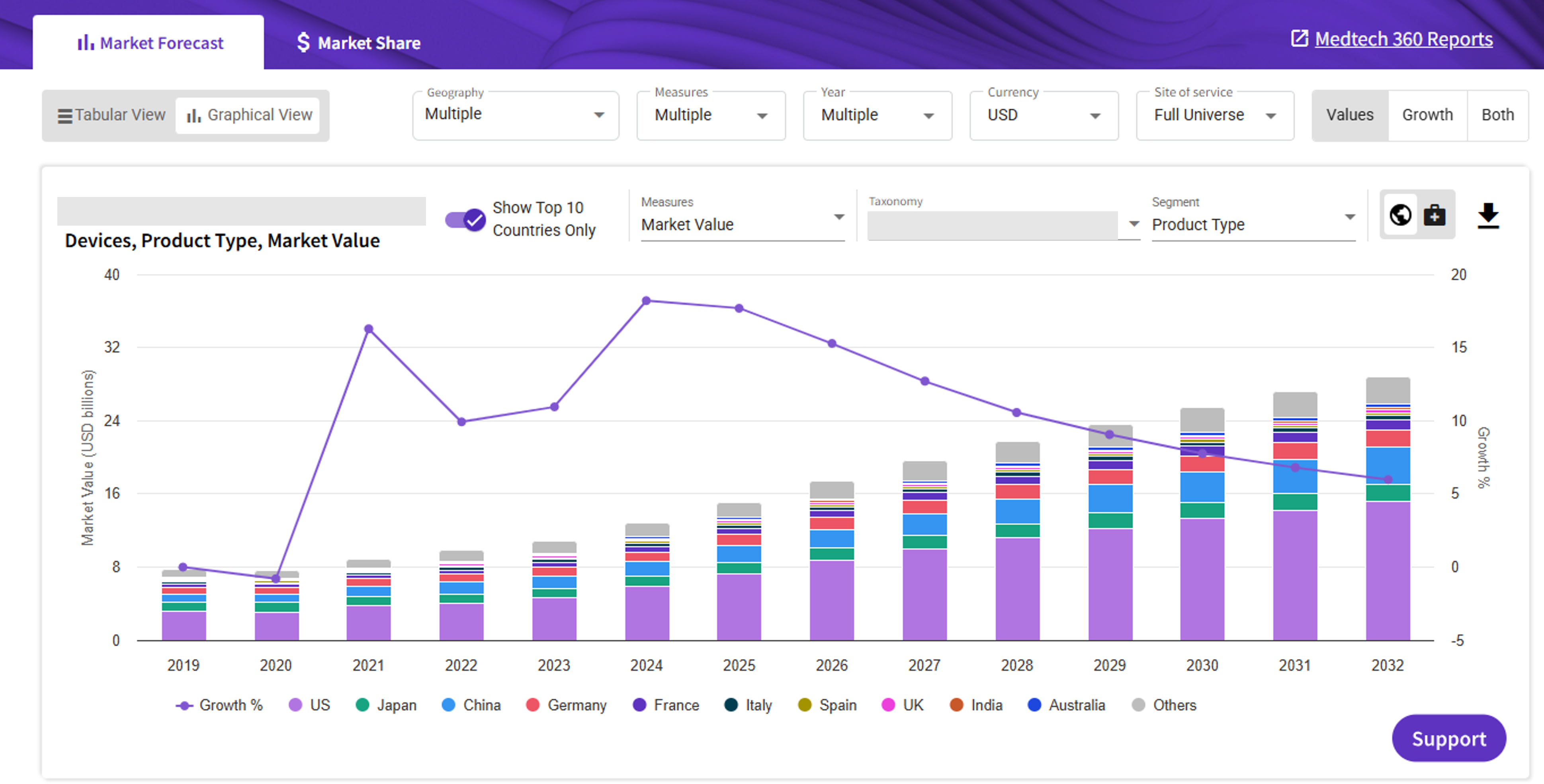
Key trends in the United States
The adoption of premium-priced devices will support US PV device market growth
-
How is the standard of care changing in PV device space as new technologies emerge?
-
How is reimbursement affecting the adoption of different devices and procedures?
-
What limitations do new technologies face in terms of approved indications?
The launch of innovative devices for treating below-the-knee lesions will bolster the US PV device market going forward
-
What are the device types and technological innovations being evaluated to address unique challenges of treating BTK lesions?
-
How will the entry new drug-eluting technologies, including BRS, affect to the adoption of endovascular techniques in the infrapop indication?
-
Which competitors are standing to benefit from new devices, and how will it shape the market going forward?
Several key players in the PV device market have expanded their presence in the US market through acquisition and partnerships
-
Which key players in the PV device market are involved in acquisitions and partnerships, and how will these developments impact key players’ shares in the future?
Continued cuts to the reimbursement rates in certain sites of service and procedure types will result in a shift in site for some procedure volumes
-
What new reimbursement updates released by the CMS will have an impact on PV device uptake?
-
How have recent reimbursement policies influenced the site-of-service penetration for different PV procedure types?

Key trends in Europe

Uptake of premium-priced devices will drive growth in the market
-
To what degree will DES and DCBs be adopted in Europe over the forecast period?
-
What is the future outlook for the adoption of covered stents?
-
How readily have IVL procedures been adopted for PV indications?
-
What impact will these trends have on the competitive landscape?
Recently published positions and guidelines from key organizations are expected to impact the adoption of paclitaxel-eluting devices
-
How will the recent FDA letter concerning paclitaxel-based devices affect device choice in Europe?
-
Will the recently MHRA published guidelines have a positive impact in the UK? And in other European countries?
-
What is the current position about paclitaxel-eluting device among physicians?
-
How will these updates affect other alternative drug-eluting devices in the market?
Procedure volume growth will be supported by increasing interventional endovascular techniques in iliac, fem-pop and infrapop indications
-
Which devices are expected to see the greatest uptake in these indications?
-
How has this focus impacted the PV device market?
M&A activities or partnerships involving key competitors may lead to shifts in market shares going forward
-
Which acquisitions have occurred, and how will these acquisitions impact competitor performance in the future?
-
How are competitors leveraging product portfolio acquisitions to expand their product offerings?
Key trends in Asia Pacific
-
PV device market will grow modestly over the forecast period, driven by the high prevalence of PAD, expanding PAD-related comorbidities, and rising adoption of premium-priced drug-eluting products.
-
Rising procedure volumes for iliac and lower limb indications and the increasing adoption of endovascular techniques over surgical intervention will also fuel market growth, particularly in China.
-
Cost constraints in publicly funded healthcare systems and limited reimbursement in the region continue to hinder the adoption of premium-priced devices and limit the pace of revenue expansion.

Key trends in Japan
The expansion of DES and DCB markets will be driven by new device launches and positive clinical data
-
What findings from ongoing and completed trials will contribute to growing use of DES and DCBs in Japan?
-
To what degree will DES and DCBs be adopted in Japan over the forecast period?
-
What strategies are competitors employing to capture sales in the growing markets for DCB and DES in Japan?
Advancements in endovascular interventions are primarily concentrating on the treatment of lower extremity conditions
-
What upcoming advancements are expected in the PV device market?
-
How are global clinical developments influencing the Japanese PV device market?
-
What new devices have been launched or are anticipated to be launched in Japan?
-
How will the introduction of IVL impact the Japanese PV device market?
-
How the expansion of IVUS for PV interventions impact market growth? Who are the competitors in this growing segment?
M&A activities or partnerships involving key competitors may lead to shifts in market shares going forward
-
Which acquisitions have occurred, and how will these acquisitions impact competitor performance in the future?
-
How are competitors leveraging product portfolio acquisitions to expand their product offerings?