Life Sciences and Healthcare
Cortellis CMC Intelligence
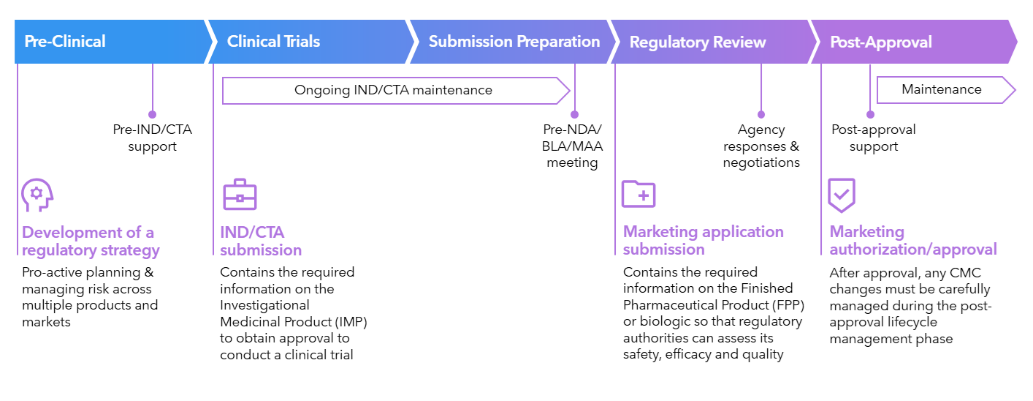
Streamline Chemistry, Manufacturing & Controls compliance across global markets
- Access comprehensive CMC requirements across 137 markets for small molecules and 64 for biologics.
- Streamline submission preparation with expert insights on general and local practice, cross-market comparisons, and eCTD Module 3-organized navigation.
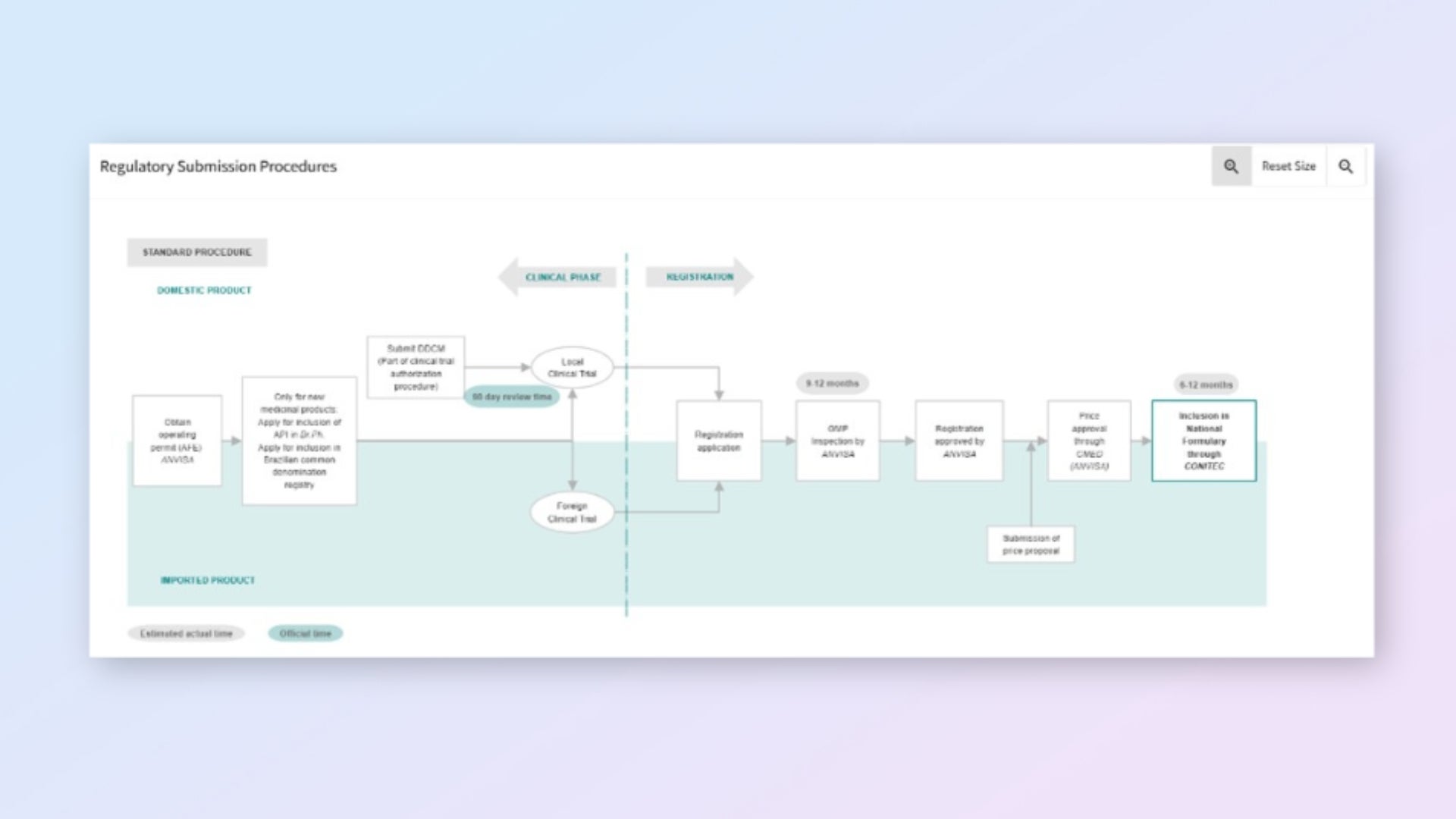
- Build realistic approval strategies with submission pathway visualizations showing official and estimated timelines.
- Never miss critical CMC regulatory changes with continuous monitoring and configurable email alerts.
Request more information
Preparing CMC submissions shouldn’t drain your resources
Cortellis CMC Intelligence centralizes Module 3 requirements for pre-approval submissions and post-approval change management across global markets. Built specifically for regulatory CMC professionals, our platform delivers specialized technical intelligence without the consultant fees.
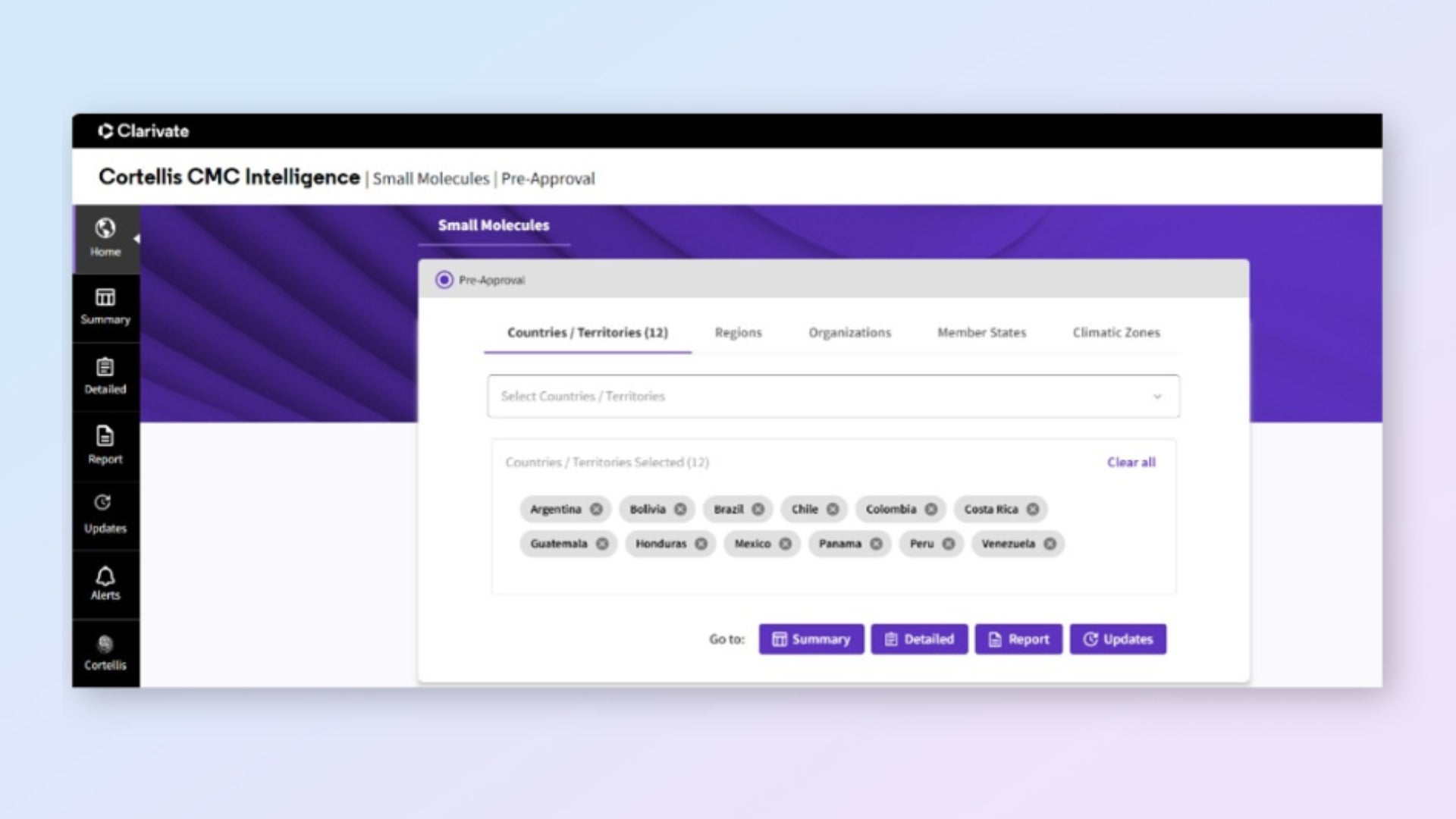
Access global CMC requirements in a single platform
Find all Module 3 requirements across 137+ markets in one centralized location. No more navigating multiple agency websites or managing scattered documents.
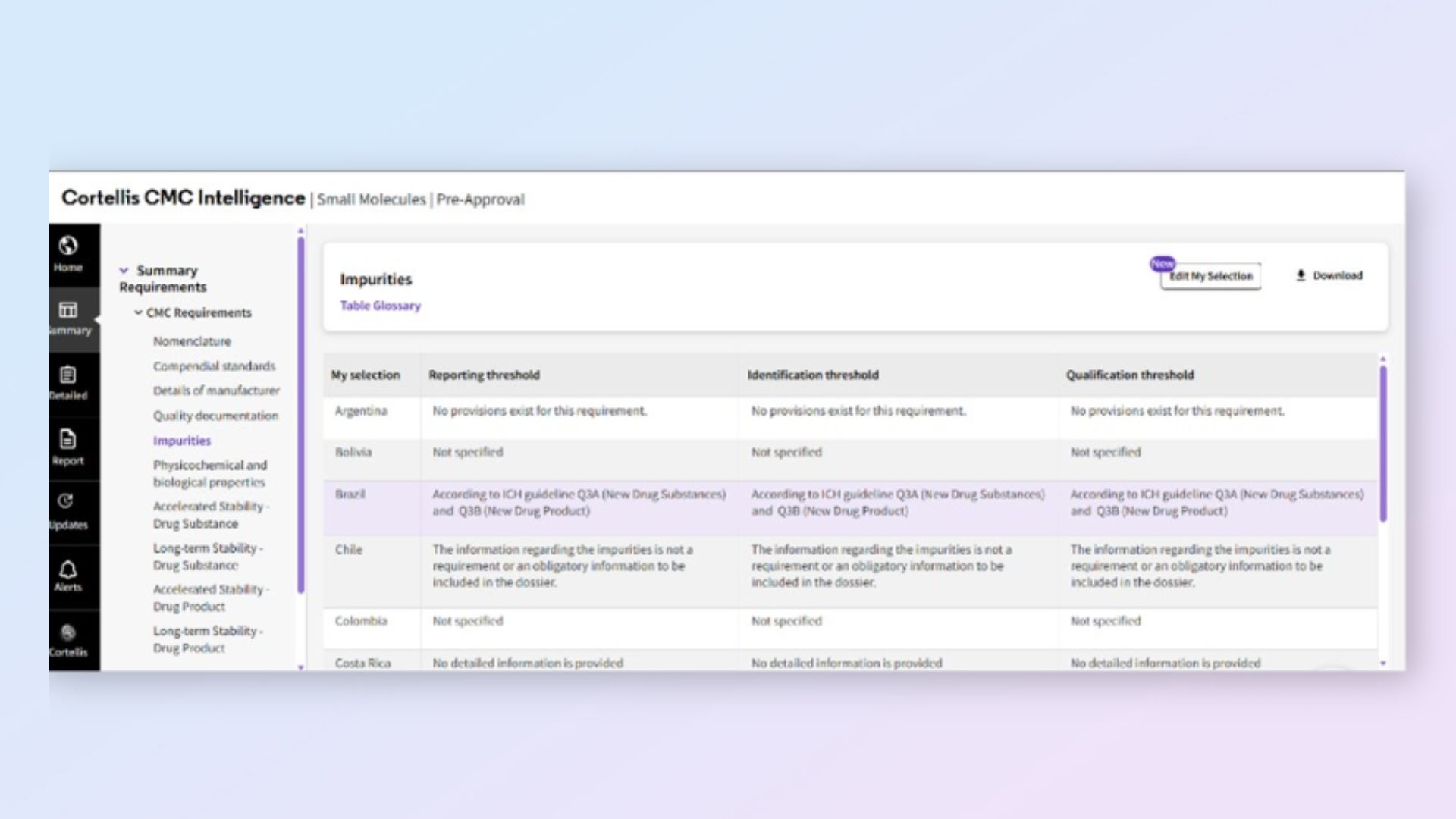
Prepare submissions for multiple markets simultaneously
Compare requirements across markets, all translated to English. Work efficiently across jurisdictions without language barriers or monitoring multiple agencies.
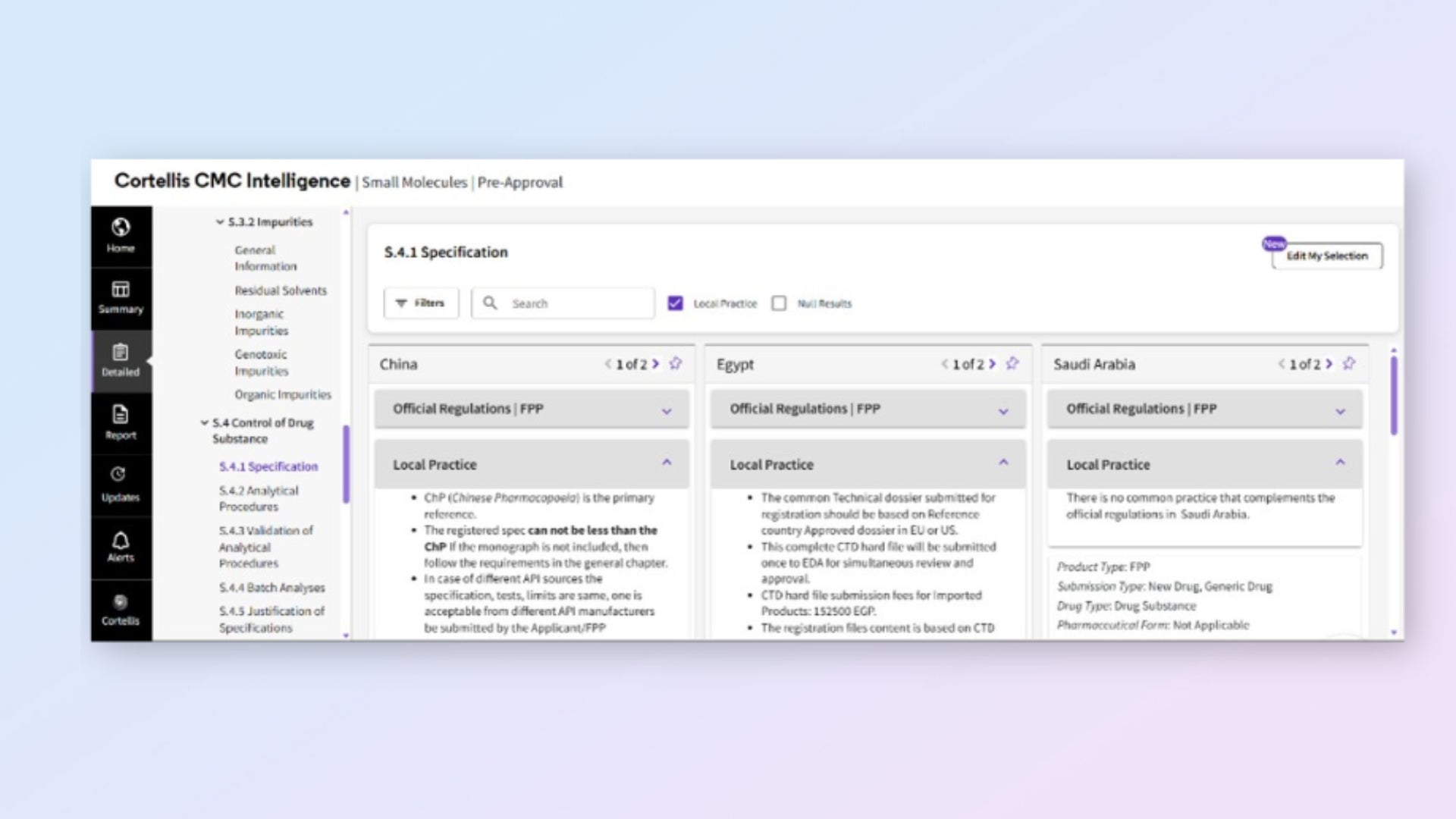
Understand local practices beyond published regulations
Access expert insights on unwritten agency expectations and local variations. Reduce reliance on consultants while improving submission quality and accuracy.
Plan with confidence using pathway visualizations
View end-to-end submission pathways with official and estimated timelines. Build realistic strategies and align stakeholder expectations.
FAQ
Cortellis CMC Intelligence is a regulatory intelligence platform designed specifically for Chemistry, Manufacturing & Controls (CMC) professionals in pharmaceutical and biotech companies. It provides centralized access to Module 3 regulatory requirements, post-approval change guidance, and expert insights across 137+ markets for small molecules and 64+ markets for biologics. Regulatory CMC managers, directors, and teams use it to prepare compliant submissions, manage post-approval changes, and stay current with evolving CMC requirements.
There are currently no other web-based CMC regulatory intelligence platforms on the market. Most pharmaceutical companies either build internal tools (like spreadsheets or databases) that quickly become outdated, or rely heavily on consulting agencies for CMC information, which can be expensive with variable quality and turnaround times. Cortellis CMC Intelligence provides a centralized, expert-curated platform specifically designed for CMC professionals, eliminating the need to piece together information from multiple agency websites in different languages or depend on costly consultants for every market entry decision.
Cortellis CMC Intelligence covers pre-approval content for 137 countries for small molecules and 64 countries for biologics across the Americas, Europe, Middle East & Africa, Asia Pacific, and Eurasia regions. This includes major markets like US (FDA), Europe (EMA), Japan (PMDA), China (NMPA), and many emerging markets. For post-approval change management, the platform covers 61 countries for both small molecules and biologics. All content is provided in English with links to original source documents, and coverage is continuously expanding.
Yes, post-approval change management is a core feature. The platform provides change classification systems and submission procedures for 61 countries covering both small molecules and biologics. This helps you navigate the complexities of managing variations across multiple markets throughout the product lifecycle.
Our global team of CMC regulatory experts continuously monitors 7,000+ source documents, updating the platform as agencies publish new guidelines, modify requirements, or issue clarifications. Users can configure email alerts for specific countries and/or requirement types to receive notifications about relevant changes. All updates include links to original source documents.